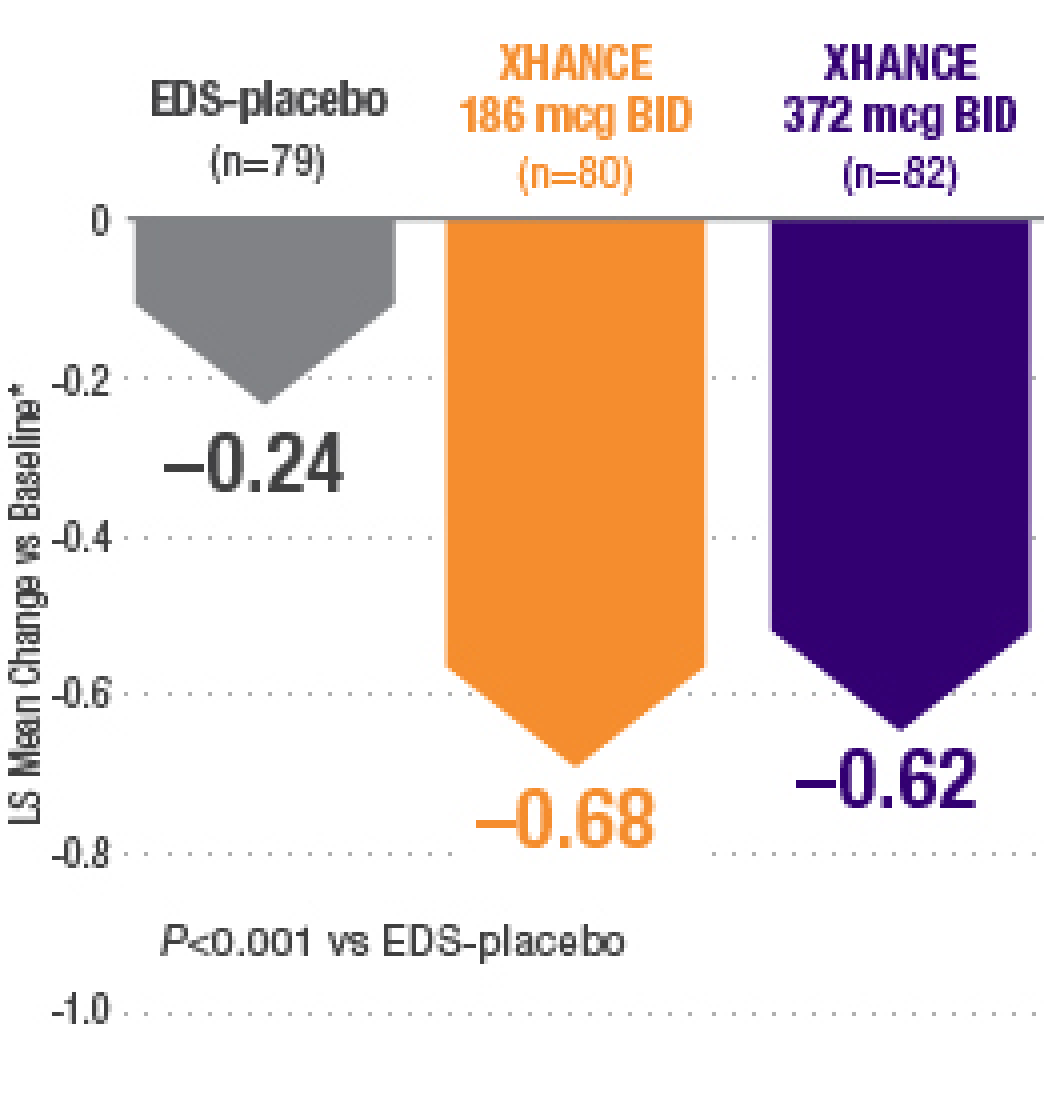
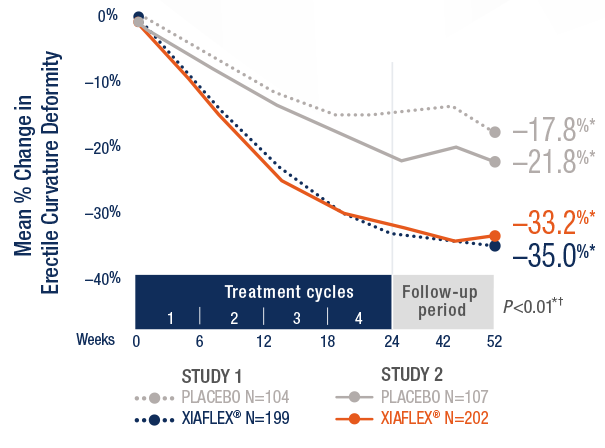
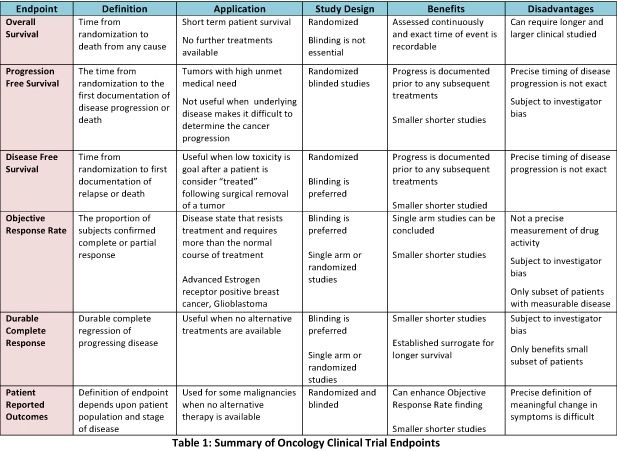
Sequential sample size re-estimation in clinical trials with multiple co-primary endpoints - WRAP: Warwick Research Archive Portal

Impact of non‐binding FDA guidances on primary endpoint selection in Alzheimer's disease trials - Yu - 2022 - Alzheimer's & Dementia: Translational Research & Clinical Interventions - Wiley Online Library
Multiple Co-primary Endpoints: Medical and Statistical Solutions A Report From the Multiple Endpoints Expert Team of the Pharmac

Defining Endpoints and Biomarkers in Inflammatory Bowel Disease: Moving the Needle Through Clinical Trial Design - Gastroenterology
Sample size determination for a specific region in multiregional clinical trials with multiple co-primary endpoints

Primary and Secondary Outcome Reporting in Randomized Trials: JACC State-of-the-Art Review | Journal of the American College of Cardiology
Multiple Co-primary Endpoints: Medical and Statistical Solutions A Report From the Multiple Endpoints Expert Team of the Pharmac